|
SGYP gained FDA approval for Trulance (plecanatide) for the treatment of adults with CIC which could pose competition to Linzess. We remind investors that recently Synergy Pharmaceuticals Inc. The company is targeting at least three additional launches of Linzess by the end of 2018. 2.2 Preparation and Administration Instructions Take Linzess on an empty stomach, at least 30 minutes prior to the first meal of the day. A dosage of 72 mcg once daily may be used based on individual presentation or tolerability. 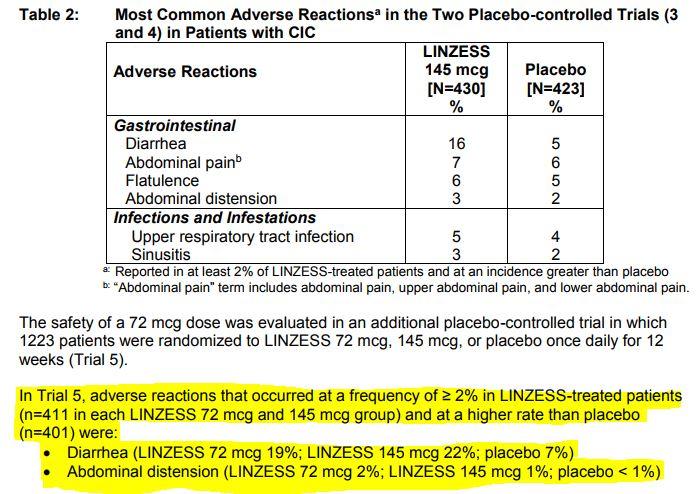 
By the end of 2020, Ironwood expects annual sales of Linzess to cross $1 billion. The recommended dosage of Linzess is 145 mcg orally once daily. Linzess has the potential to garner blockbuster sales. Linaclotide is used to treat certain types of bowel problems (irritable bowel syndrome with constipation, chronic idiopathic constipation). Ironwood has partnered with Astellas Pharma for the development and commercialization of Linzess in Japan and with AstraZeneca plc AZN in China, Hong Kong and Macau. The photos shown are samples only Not all photos of the drug may be displayed. Subsequently, in Jun 2016, the FDA accepted the companies’ sNDA for a 72 mg dosage of Linzess for review. Data from the study showed that patients when treated with Linzess (72 mcg) demonstrated a statistically significant improvement compared to placebo on the 12-week complete spontaneous bowel movements (CSBM) overall responder endpoint, thereby meeting the primary endpoint of the study. The stock lost 2.7% during the period, while the industry registered a decline of 5.3%.Īpproval of the new dose was supported by a phase III clinical trial on 1,223 adult patients with CIC. Shares of Ironwood have outperformed the Zacks classified Medical-Drugs industry in the last six months. In Oct 2015, Allergan acquired exclusive rights for the development and commercialization of Linzess in the EU. Note that Linzess is marketed by Allergan for IBS-C in Europe under the brand name Constella. Approval of a lower dose would provide patients with an additional treatment option. In the IBS-C indication, it is approved as a 290 mcg capsule to be taken once daily. The drug is approved in the CIC indication as a 145 mcg capsule to be taken once daily. for the treatment of adults with irritable bowel syndrome with constipation (IBS-C) or CIC. So, doctors have the flexibility to give you a higher or lower dose after evaluating your condition. 
Secondly, the trulance is available in 3mg dose only, whereas the linzess come in 3 doses including 72, 145, 290 mcg capsules. Linzess, Ironwood's sole marketed product, is available now in the U.S. First off, you can take trulance with or without food in your stomach which you can’t do with linzess. People can save money on Linzess 72 mcg by comparing Linzess prices at Canadian and international online pharmacies with discount prices available at U.S. The generic and brand version are available at accredited international online pharmacies. The lower dose is expected to be available in the U.S. Linzess is available only as a brand version in the U.S. IRWD and partner Allergan plc AGN announced that the FDA has approved a lower dose strength of Linzess (linaclotide) – 72 mcg – for the treatment of adult patients with chronic idiopathic constipation (CIC).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
